
A tailored phase I-specific patient-reported outcome (PRO) survey to capture the patient experience of symptomatic adverse events | British Journal of Cancer
![PDF] Patient-reported outcomes and the evolution of adverse event reporting in oncology. | Semantic Scholar PDF] Patient-reported outcomes and the evolution of adverse event reporting in oncology. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/72d6066aa43136e48ae6226e3dba7e542a7a02de/5-Table1-1.png)
PDF] Patient-reported outcomes and the evolution of adverse event reporting in oncology. | Semantic Scholar
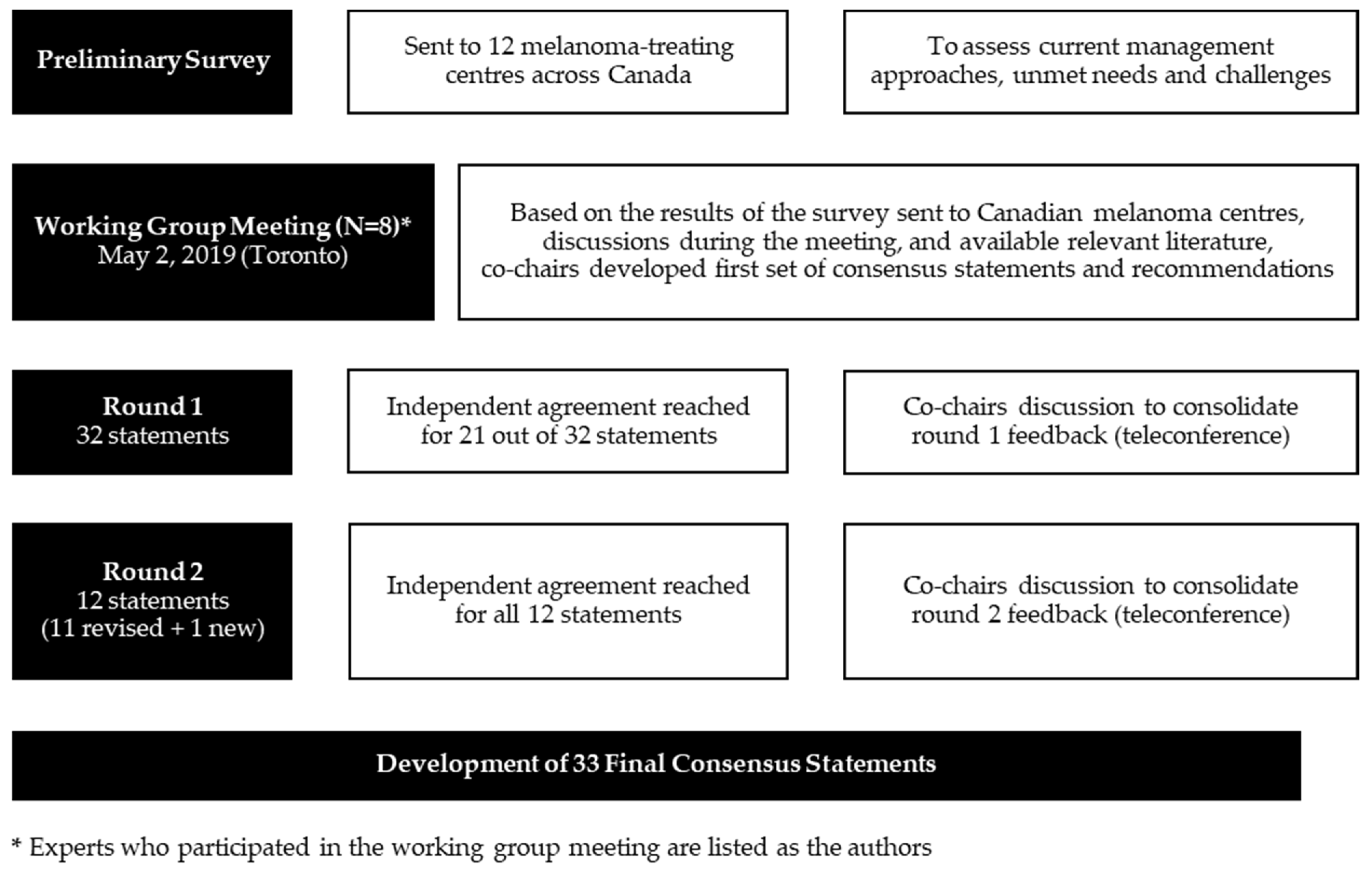
Current Oncology | Free Full-Text | Management of Pyrexia Associated with the Combination of Dabrafenib and Trametinib: Canadian Consensus Statements

Feasibility of Implementing the Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events in a Multicenter Trial: NCCTG N1048 | Journal of Clinical Oncology
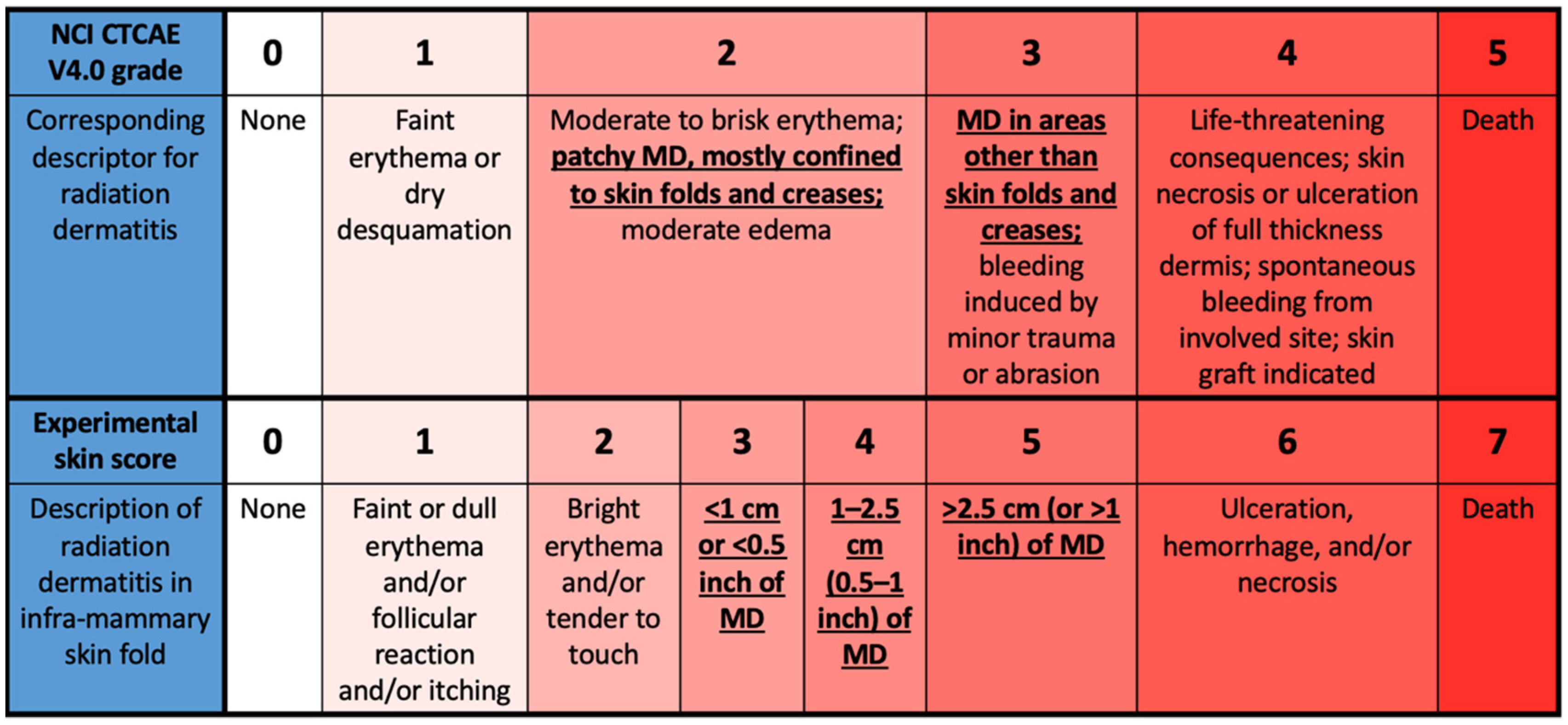
Current Oncology | Free Full-Text | Validation of a Patient-Reported Outcome Measure for Moist Desquamation among Breast Radiotherapy Patients

Feasibility of Implementing the Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events in a Multicenter Trial: NCCTG N1048 | Journal of Clinical Oncology

Annals of Surgery on X: "Patient-Reported Adverse Events during Neoadjuvant Therapy in a Phase 2 Borderline Resectable Pancreatic Cancer Clinical Trial (Alliance A021501) #AmerSurg23 https://t.co/zoJ0tFPrHB" / X
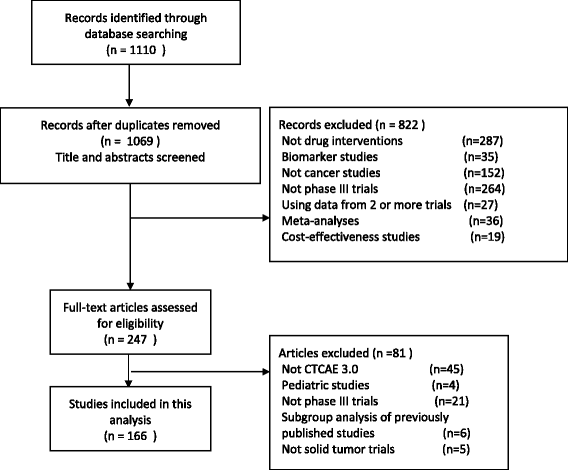
Use and misuse of common terminology criteria for adverse events in cancer clinical trials | BMC Cancer | Full Text

![Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF](https://image.slidesharecdn.com/ctcae4-022009-09-15quickreference8-5x111-120711021618-phpapp01/85/ctcae-402-20090915quickreference85x111-2-320.jpg?cb=1668437655)


![Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF](https://image.slidesharecdn.com/ctcae4-022009-09-15quickreference8-5x111-120711021618-phpapp01/85/ctcae-402-20090915quickreference85x111-3-320.jpg?cb=1668437655)






![Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF](https://image.slidesharecdn.com/ctcae4-022009-09-15quickreference8-5x111-120711021618-phpapp01/85/ctcae-402-20090915quickreference85x111-4-320.jpg?cb=1668437655)
![Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF Ctcae 4.02 2009-09-15_quickreference_8.5x11[1] | PDF](https://image.slidesharecdn.com/ctcae4-022009-09-15quickreference8-5x111-120711021618-phpapp01/85/ctcae-402-20090915quickreference85x111-19-320.jpg?cb=1668437655)


